Indications and Limitation of Use: ADYNOVATE is indicated for use in adult and pediatric patients with hemophilia A (congenital factor VIII deficiency) for on-demand treatment and control of bleeding episodes, perioperative management, and routine prophylaxis to reduce the frequency of bleeding episodes. ADYNOVATE is not indicated for the treatment of von Willebrand disease.1
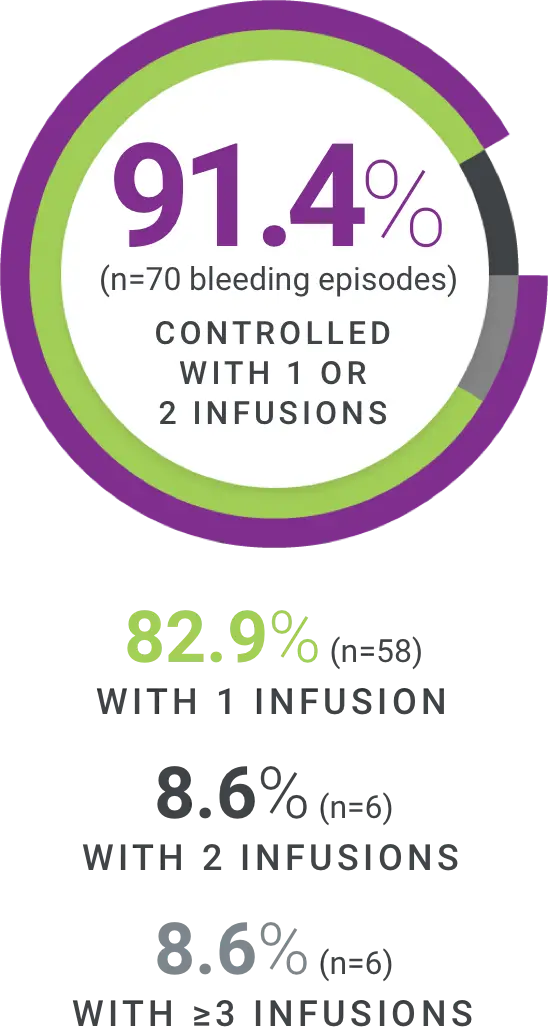
†Excellent defined as full relief of pain and objective signs of bleeding cessation; Good defined as definite pain relief and/or improvement in signs of bleeding.
CONTRAINDICATIONS: ADYNOVATE is contraindicated in patients who have had prior anaphylactic reaction to ADYNOVATE, to the parent molecule (ADVATE [Antihemophilic Factor (Recombinant)]), mouse or hamster protein, or excipients of ADYNOVATE (e.g. Tris, mannitol, trehalose, glutathione, and/or polysorbate 80).
See Detailed Important Risk Information below.
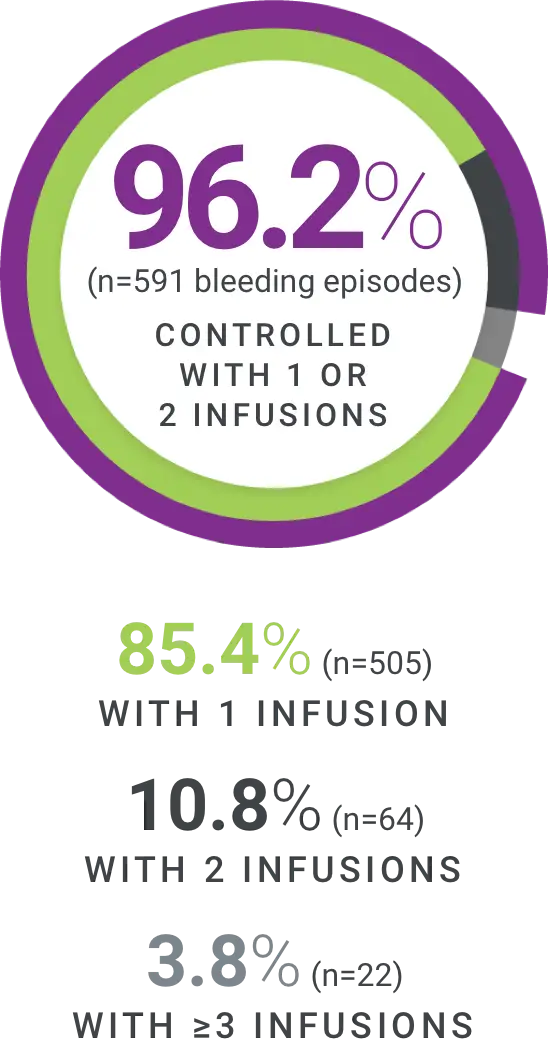
†Excellent defined as full relief of pain and objective signs of bleeding cessation; Good defined as definite pain relief and/or improvement in signs of bleeding.
Hypersensitivity Reactions: Hypersensitivity reactions, including anaphylaxis, have been reported with ADYNOVATE. Hypersensitivity reactions that can progress to anaphylaxis may include angioedema, chest tightness, dyspnea, wheezing, urticaria, pruritus, and nausea and vomiting. Immediately discontinue administration and initiate appropriate treatment if hypersensitivity reactions occur.
See Detailed Important Risk Information below.


Actual ADYNOVATE patient
Individual results may vary.
Brett, 41 years old
My Hemophilia A Story
My Experience with ADYNOVATE
The Reasons I Want to Stay on ADYNOVATE
*Based on units sold as of August 2024.
Formation of neutralizing antibodies (inhibitors) to FVIII can occur following administration of ADYNOVATE. Monitor patients regularly for the development of FVIII inhibitors by appropriate clinical observations and laboratory tests.1


The efficacy of ADYNOVATE for prophylaxis was studied in clinical trials
Get the results
ADYNOVATE has a twice-weekly dosing regimen that you can tailor to each patient.1
See HowCONTRAINDICATIONS: ADYNOVATE is contraindicated in patients who have had prior anaphylactic reaction to ADYNOVATE, to the parent molecule (ADVATE [Antihemophilic Factor (Recombinant)]), mouse or hamster protein, or excipients of ADYNOVATE (e.g. Tris, mannitol, trehalose, glutathione, and/or polysorbate 80).
You are being directed to ADYNOVATEPro.com. The information
on this website is intended for U.S. healthcare professionals only.